September 10, 2022
Most biology textbooks have opening chapters that evaluation chemistry. I typically inform my college students that we have to study slightly little bit of chemistry as a result of actually, organisms are simply luggage of chemical substances. Superior courses might want to perceive some fundamentals of chemistry: molecular bonds, pH scale, and water chemistry.
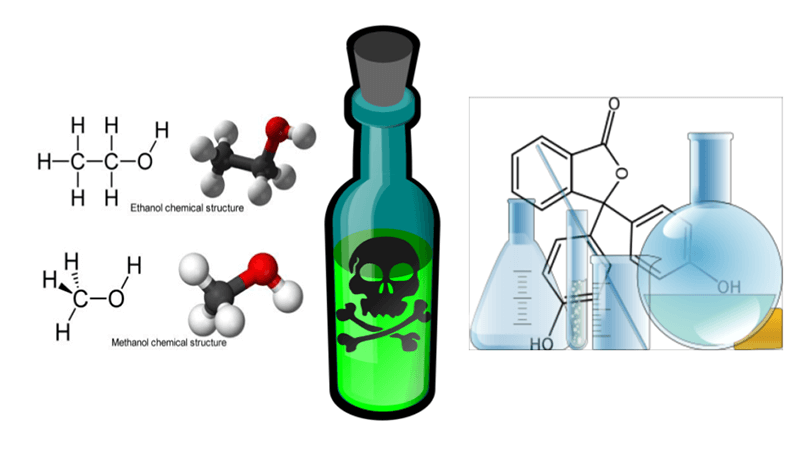
This text with questions compares two natural molecules, ethanol and methanol. Ethanol is the principle ingredient in alcohol drinks, created from the fermentation of grains and fruits. Methanol has a really related chemical construction, with just one carbon in its spine fairly than the 2 present in ethanol. Each have an alcohol (OH) group connected.
Throughout Prohibition, bootleggers typically created a model of “spirits” from methanol by burning wooden. Methanol is a poison! The outcome was that individuals ingesting methanol would die or turn out to be blind as methanol was transformed right into a extra poisonous compound: formic acid. This course of is called toxification.
College students learn the article that describes the compounds and their results whereas answering associated questions. A closing part describes one other sort of poisoning from Lindol. Alcohol spiked with this compound might result in paralysis, a situation known as “Jake Leg” after the identify of the alcohol that was poisonous (Ginger Jake.)